Vaccine technologies for the battle against COVID-19 pandemic
Avertim has been working in the Life Sciences sector for more than a decade and some of our clients include leading vaccines development and manufacturing companies which we have supported across their entire life cycle of local and global projects. Due to this background it goes without saying that we are watching the emergence of new vaccines technologies with great interest. Let us have a quick overview here.
Challenges
Global healthcare is facing multiple challenges raised by global population increase, travelling habits, demographic change as well as through human’s influence on environment and climate. Highly concentrated urban settings and intensively travelling all around the world together with extreme increase of contact between people favor the rapid spreading of the ongoing pandemic. Traditional vaccine development techniques using established methods would require up to 10 years until the launch of a new product. However, new vaccine technologies can help to promote the development of a new vaccine with immunogenicity and long period of effectiveness.
Technologies used for COVID-19 vaccine development
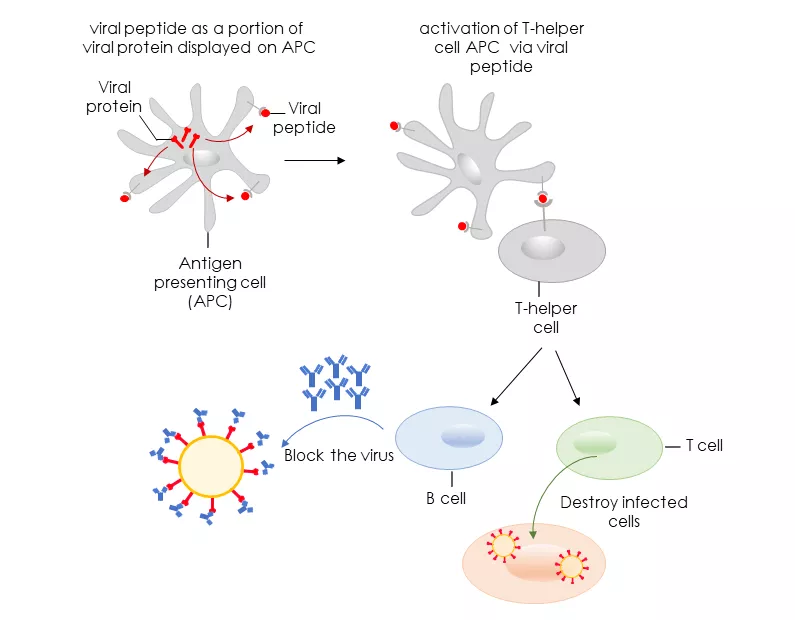
Figure 1: Immune response
The aim of COVID-19 vaccine development is to identify suitable viral antigens with the ability to induce immunogenicity (ability of a foreign substance, such as an antigen, to provoke an immune response in the body) in humans in order to prevent diseases caused by pathogens. The human immune system can learn to recognize the real virus via engineered antigens that are taken up at the beginning by antigen-presenting cells (APCs). Thereby, APCs show a part of the viral antigens on their cell surface and thus activate T-helper cells. T helper cells activate B cells to block viruses and T cells to destroy infected cells, as shown in figure 1.
Virus vaccines, viral-vector vaccines, protein-based vaccines, and nucleic-acid vaccines are the main techniques used by researchers across the world to develop vaccines against COVID-19 (see figure 2).
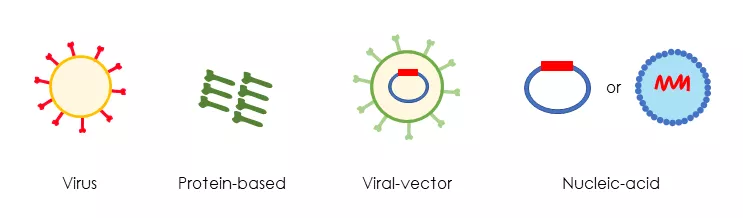
Figure 2: Different vaccine technologies
Virus vaccines are conventional vaccines developed by weakening or inactivating the whole pathogen. Weakened viruses can be identified and selected as candidate for virus vaccines if they pick up mutations which make them less pathogenic (i.e. cause less severe forms of a disease). Alternatively, inactivated viruses can be generated using heat or chemicals. Disadvantages of these methods are that they require large amounts of infectious virus, it is difficult to find right conditions to cultivate viruses, and unfavorable for highly infectious or uncharacterized pathogens. Furthermore, specialized labors with adequately high biosafety levels are required for virus cultivation. In the end, after inactivation of the virus it may no longer induce protective immune responses.1,2
Another approach is represented by viral-vector vaccines which are classified into replicating viral vectors and non-replicating vectors based on their ability to replicate after vaccine injection (see figure 3). They are genetically engineered viruses which are already weakened so that they cannot cause diseases. The Ebola vaccine is an example of a replicating viral vector, which shows strong immune response and tends to be safe.3 Although non-replicating vectors cannot replicate in the cell they induce long-lasting immunity. Advantages of this technology are that a large amount of different viral vectors is well-known and that their genome can be easily manipulated to produce any antigen of choice. However, viral vectors are genetically modified, and therefore present a potential risk to human health and environment. Such risks must be carefully assessed before entry into and during clinical studies.2,4
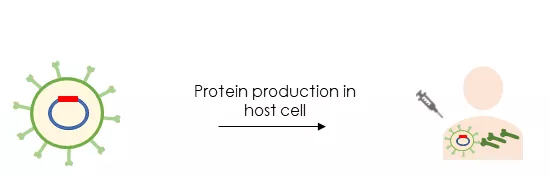
Figure 3: Viral-vector vaccines
Next approach is protein-based vaccines composed of viral protein subunits and virus-like particles. Protein subunits vaccines are produced using biologically engineered systems in yeast or bacteria (see figure 4). Similar vaccines against SARS virus have protected monkeys against infection but have not been tested on people. However, adjuvants need to be used to improve the presentation of viral proteins to the immune system and to enhance the immunogenicity. Virus-like particles are known as empty virus shells mimic the coronavirus structure. But they cannot cause infection because they lack genetic material. They can trigger strong immune response, but they are difficult to produce. 2,5
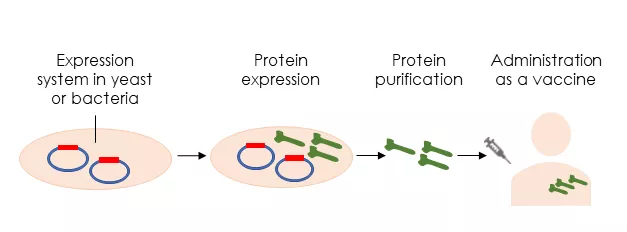
Figure 4: Protein-based vaccines
The newest vaccine technology is nucleic acid vaccines which only use the genetic material of virus either DNA or RNA (see figure 5). These molecules are injected inside the body, which will produce viral protein and trigger an immune response. Nucleic acid-based vaccines are safe and easy to develop without using the whole virus. But till now no licensed vaccines use this approach.2,6,7
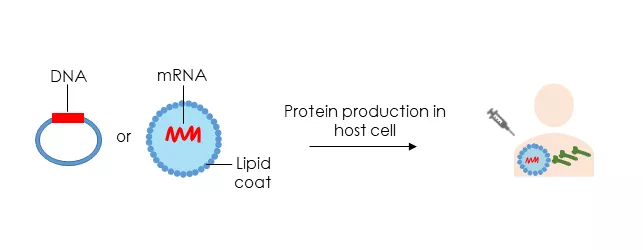
Figure 5: Nucleic acid vaccines
The run for vaccines against COVID-19
Obviously, established methods are no longer sufficient to counteract outbreak situations of the pandemics to identify a new vaccine candidate. In April 2020, more than 90 vaccines against COVID-19 have been developed worldwide. At least 20 teams develop vaccines using nucleic acids, while 25 groups work on viral vector vaccines. 33 teams work on protein-based vaccines and only seven teams develop vaccines using virus.9,10 Moderna is the first pharmaceutical company entered the clinical study, where it works on mRNA vaccines. Meanwhile, Pfizer teams up with BioNTech to develop the promising mRNA vaccines. While University of Oxford and AstraZeneca are developing non-replicating viral-vector based vaccines, Sanofi and GSK are working on recombinant protein vaccines.11
In the next article with the title "166 vaccines in the run against COVID-19 - Germany strongly positioned” you get more updates of the vaccines being currently developed against COVID-19.
You want to learn more about Avertim’s activities in the Life Sciences industry? Get in touch with us here
References
- Morabito, K. M. & Graham, B. S. Zika Virus Vaccine Development. J. Infect. Dis. 216, S957–S963 (2017).
- Callaway, E. The race for Coronavirus vaccines. Nature 580, 576–577 (2020).
- Ledgerwood, J. E. et al. Chimpanzee Adenovirus Vector Ebola Vaccine. N. Engl. J. Med. 376, 928–938 (2017).
- Ramezanpour, B., Haan, I., Osterhaus, A. & Claassen, E. Vector-based genetically modified vaccines: Exploiting Jenner’s legacy. Vaccine 34, 6436–6448 (2016).
- Draper, S. J. et al. Recent advances in recombinant protein-based malaria vaccines. Vaccine 33, 7433–7443 (2015).
- Williams, J. Vector Design for Improved DNA Vaccine Efficacy, Safety and Production. Vaccines 1, 225–249 (2013).
- Brito, L. A. et al. Self-Amplifying mRNA Vaccines. Advances in Genetics vol. 89 (Elsevier Ltd, 2015).
- Thompson, S. A. How Long Will a Vaccine Really Take? https://www.nytimes.com/interactive/2020/04/30/opinion/coronavirus-covi…
- Thanh Le, T. et al. The COVID-19 vaccine development landscape. Nat. Rev. Drug Discov. 19, 305–306 (2020).
- WHO. Draft landscape of COVID-19 candidate vaccines – 20 April 2020. https://www.who.int/blueprint/priority-diseases/key-action/novel-corona….
- 9 Pharmaceutical Companies Racing For A COVID-19 Vaccine. https://www.forbes.com/sites/moneyshow/2020/06/16/9-pharmaceutical-comp….
