Agile project management is well known-for its success in software. The pharmaceutical industry is also increasingly employing Agile Approaches to navigate complex regulations and power business improvement.
At Avertim, we partnered with several pharmaceutical clients to successfully deploy Agile Approaches in their organisation.
Here's our summary of the benefits and some of its best uses.
Why is Agile project management becoming increasingly popular in Pharma ?
Agile's iterative approach to project management is proven and popular, with around 71% of U.S companies employing it to drive innovation and stay competitive.
While it is well-known as a dominating for in software development, agile project management (Agile PM) is increasingly making its benefits seen in the pharmaceuticals industry. Depending on complexity, and correctly implemented, Agile PM can be a far more efficient way to run projects.
Agile Project Management and Pharma's unique challenges.
The pharmaceutical industry faces many challenges, including increased competition, complex regulations, and the need for faster drug development and production.
Operating in a highly regulated environment to ensure patient safety and product quality, the sector is known for its strict regulations and complex processes. This has been viewed as a hurdle to implementing agile practices.
However, when properly implemented, agile project management is highly effective at enabling pharmaceutical organisations to both navigate regulation and drive business improvement.
Why the Agile approach fits in the pharmaceutical industry
Agile project management increases transparency. By maintaining clear documentation, tracking changes, and involving stakeholders at each stage, organisations ensure regulatory compliance while embracing agile practices. This enhances visibility and responsability throughout the entire development process.
Agile strongly supports effective risk management and compliance. By proactively incorporating risk assessment and mitigation measures into each iteration, companies can ensure products meet regulatory standards.
This particularly empowers organizations to respond quickly to regulatory change. The iterative development approach enables teams to adapt quickly and to implement essential modifications. This means organisations can ensure compliance and respond quickly to changing regulatory needs.
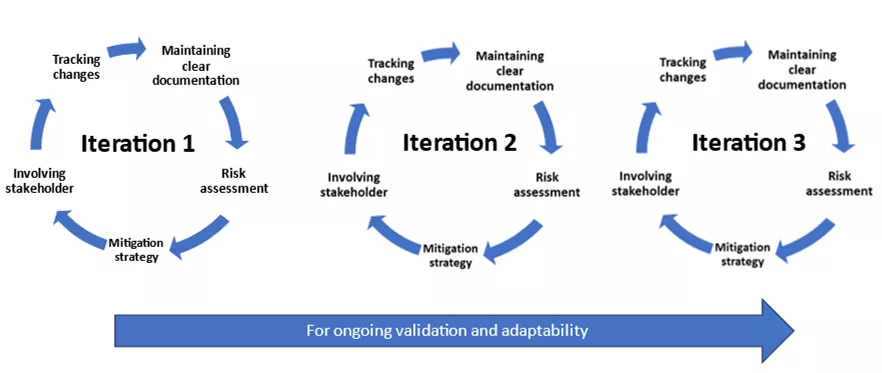
Some of the best use cases for Agile in the pharmaceutical industry.
1. New Product Development:
Agile techniques manage the complex process of developing new products by dividing development process into smaller iterations. Pharmaceutical teams can validate hypotheses, incorporate regulatory feedback, and make necessary adjustments, in these smaller iterations.
This leads to faster time-to-market results while ensuring regulatory compliance.
2. Technology Implementation:
New technologies, such as Manufacturing Execution Systems (MES), Laboratory Information Management Systems (LIMS) or Computer Systems Validation (CSV) can be implemented with agile methodologies. Agile methodologies promote close collaboration among IT, operations and regulatory teams. This helps technology fit in smoothly, ensure compliance, and keeps projects on track.
Here's one example of how Avertim helped a major pharmaceutical company in their digitalisation journey.
3. Process Optimisation:
Agile methodologies ensure process optimisation initiatives are effective. Through techniques such as value stream mapping and rapid prototyping, pharmaceutical manufacturers can pinpoint bottlenecks, implement process improvements, and enhance operational efficiency while adhering to Good Manufacturing Practice (GMP) guidelines.
Integrating Agile PM in pharmaceutical companies
When implemented successfully, Agile's iterative approach to project management is highly effective at driving improvement even in highly regulated industries. It can be highly useful in handling the complex regulatory landscape of the pharmaceutical industry while focusing on business improvement.
There is perception that it is too challenging to introduce but many pharmaceutical companies have already done so successfuly - often by partnering with a team who understand the industry's specific needs. By embracing agile methods and taking advantage of new technology, pharmaceutical companies can build a culture of continuous improvement in a changing industry.
Want a partner who understands your challenges?
Avertim's internal Centre of Excellence for project, portfolio and program management has extensive experience in the pharmaceutical industry.
Our expert understand how to deliver this methodology in the most effective way.


